Clinical Application of Liquid Biopsy-Based Circulating Tumor DNA for Follow-Up Care in Breast Cancer Management: Lessons from Other Solid Cancers
DOI:
https://doi.org/10.15419/bmrat.v10i3.797Keywords:
Circulating tumor DNA, liquid biopsy, breast cancer managementAbstract
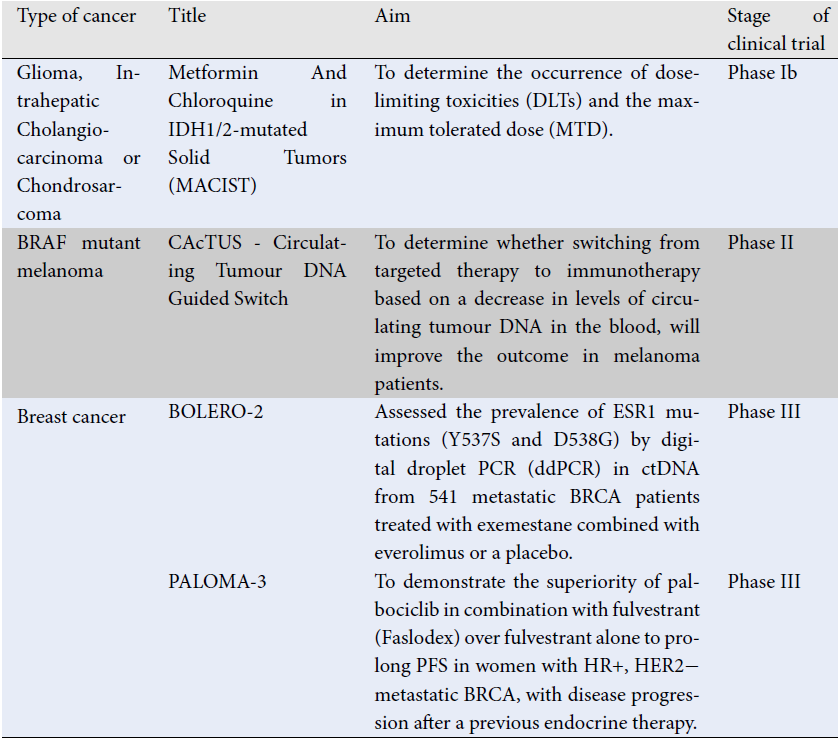
Early detection of relapse following successful curative-intent breast cancer surgery is an essential strategy for survival. Unfortunately, conventional screening via radiological imaging and tissue biopsies may be confounded by tumor size. Moreover, tissue biopsy is comparably invasive, clinically challenging, and predisposes medical and mental health complications. The use of liquid biopsies for postoperative screening in breast cancer survivors has been explored as a safer and easier alternative. The increasing popularity of the minimally invasive approach is due to its simplicity in accessing and obtaining the analyte and its lower associated costs. Using circulating tumor DNA (ctDNA) as a novel marker, the method enables real-time monitoring of disease-free survival and detection of potential cancer recurrence through cancer-specific alterations in ctDNA release from cancer cells and its short half-life. The promising clinical applications of ctDNA have fueled interest in developing new biomarkers with superior sensitivity for earlier detection of cancer recurrence, leading to the improved efficacy and efficiency of treatments and management. However, establishing this less invasive screening tool remains a challenge. In this review, we discuss the state-of-the-art in ctDNA-based liquid biopsies with a focus on the recent progress, challenges, and future directions of this technique for clinical applications in the follow-up care of breast cancer survivors.
Published
Issue
Section
License
Copyright The Author(s) 2017. This article is published with open access by BioMedPress. This article is distributed under the terms of the Creative Commons Attribution License (CC-BY 4.0) which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
