Different concentrations of nivolumab reduce PD-1 expression but not tumour growth in an EMT6 mouse model
DOI:
https://doi.org/10.15419/bmrat.v11i2.864Keywords:
PD-1, EMT6, Nivolumab, Mouse-bearing tumour modelAbstract
Introduction: The interaction between the T cell immune checkpoint proteins, the programmed death-1 (PD-1) receptor, and its ligand PD-L1 plays a crucial role in T cell suppression and the evasion of cancer cells from immune detection, thereby promoting tumour growth. Nivolumab, a PD-1 inhibitor, disrupts this interaction, offering a potential therapeutic anti-cancer strategy. The goals of this study were to identify the optimal dosage of nivolumab that effectively decreases PD-1 protein expression in a mouse model, and to examine the impact on tumour growth.
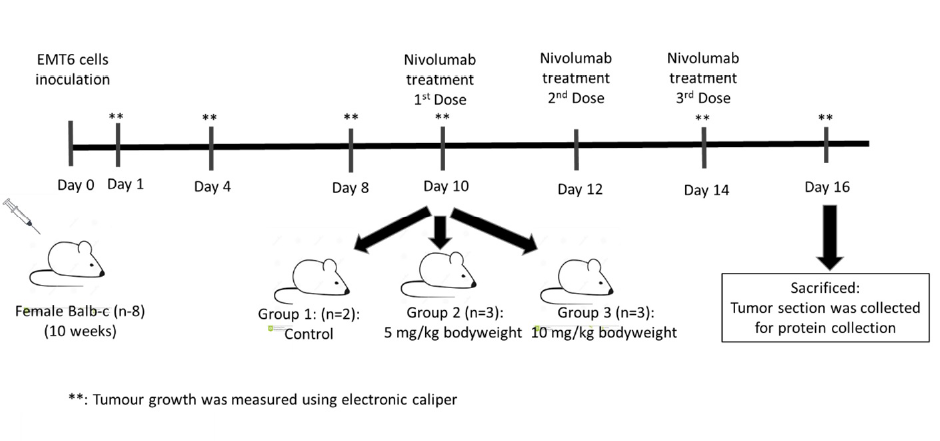
Methods: We utilized a xenograft mouse model with EMT6 mammary carcinoma cells. Eight female Balb/C mice were inoculated with EMT6 cells and assigned to three groups: a control (n = 2) and two treatment groups receiving nivolumab at 5 mg/kg (n = 3) and 10 mg/kg (n = 3) on days 10, 12, and 14 postinoculation. Tumour sizes were measured at specified intervals using electronic callipers, and the mice were sacrificed on day 16 to assess PD-1 protein levels via sandwich ELISA.
Results: There was no significant difference in tumour volume across all groups compared to the controls. PD-1 protein expression was significantly lower in Group 3 (10 mg/kg nivolumab) than in both Group 2 and the control group.
Conclusion: Nivolumab administration at a dose of 10 mg/kg markedly reduced PD-1 protein expression in a tumour-bearing mouse model, suggesting that higher doses of nivolumab may be more effective in modulating immune responses against tumour growth. These findings contribute to our understanding of nivolumab's pharmacodynamics and underscore the importance of dose optimization in enhancing the therapeutic efficacy of the drug against cancer.
Published
Issue
Section
License
Copyright The Author(s) 2017. This article is published with open access by BioMedPress. This article is distributed under the terms of the Creative Commons Attribution License (CC-BY 4.0) which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
