Effects of fenugreek seed powder on enteral nutrition tolerance and clinical outcomes in critically ill patients: A randomized clinical trial
DOI:
https://doi.org/10.15419/bmrat.v5i7.462Keywords:
Enteral nutrition tolerance, Fenugreek seeds, Gastric residual volume, Intensive care unitAbstract
Background: Enteral nutrition (EN) intolerance is a common complication in critically ill patients that contributes to morbidity and mortality. Based on the evidence of curing effects of fenugreek seeds in some gastrointestinal disorders, this study aimed to determine the effects of fenugreek seed powder on enteral nutrition tolerance and clinical outcomes in critically ill patients.
Materials & Methods: A randomized, double-blinded clinical trial of 5-day duration was conducted on 60 mechanically ventilated patients divided in 2 groups (n=30). Group 1 was given fenugreek seed powder by gavage, twice a day in addition to routine care, while Group 2 received only routine care. Enteral nutrition tolerance and clinical outcomes were measured throughout the study. Demographic and clinical data were recorded and clinical responses to the primary outcome (enteral nutrition tolerance) and secondary outcome (other clinical factors) were interpreted. Data were analyzed using the independent t-test, Chi-squared test, covariance analysis, and repeated measure ANOVA via SPSS statistical software (v. 20); statistical significance was set at p< 0.05.
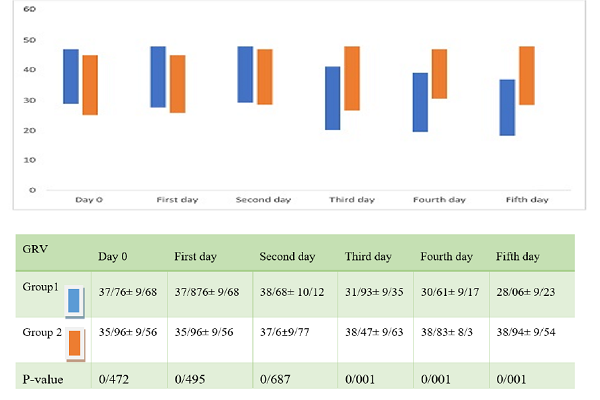
Results: Patients who were fed with the fenugreek seed powder showed a significant improvement in enteral nutrition tolerance, as well as some complications of mechanical ventilation for Group 1, as compared with Group 2. The mortality rates were not different between the two groups.
Conclusion: This study shows the beneficial effects of fenugreek seeds on food intolerance in critically ill patients and that the seed powder can be used as an add-on therapy with other medications. Thus, the use of fenugreek seeds to treat mechanically ventilated patients is recommended.
Downloads
Published
Issue
Section
License
Copyright The Author(s) 2017. This article is published with open access by BioMedPress. This article is distributed under the terms of the Creative Commons Attribution License (CC-BY 4.0) which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
